From the Microscope to Molecular Maps How spatial biology is redefining pathology

By Professor Paul Murray, Head of Department of Pathology and Lead of Viral Immunology Research Group
For more than a century, pathology has relied on the microscope. By examining stained tissue sections on glass slides, pathologists have been able to diagnose and classify diseases and guide treatment decisions. While this traditional approach remains fundamental to medical practice, advances in molecular technologies are transforming how we understand tissue biology. Increasingly, pathology is evolving from static histological interpretation toward detailed molecular maps of tissues, revealing how cells interact within their local environment.
This transformation is being driven by the rapidly developing field of spatial biology. Spatial biology integrates advanced imaging technologies with genomics (the study of an organism's entire set of genes) and proteomics (the study of all proteins within an organism) to analyse tissues while preserving their spatial organisation. So, instead of simply identifying which cells are present, researchers can now determine where cells are located, how they interact with neighbouring cells and how molecular signals vary across different regions of a tissue.
In cancer, this spatial perspective is particularly important. Tumours are not simply collections of malignant cells; they are complex ecosystems composed of cancer cells, immune cells, blood vessels and structural components such as the extracellular matrix. The organisation of the non-cancerous elements, often referred to as the tumour microenvironment, plays a critical role in determining how cancers grow, evade the immune system and respond to therapy. Work being done at RCSI Medical University of Bahrain using spatial biology is already helping us understand why some virally-induced cancers, including highly aggressive malignancies like diffuse large B-cell lymphoma (a fast-growing type of blood cancer in the lymphatic system), are able to evade immune surveillance.
Such insights are particularly important as modern cancer treatments increasingly target the immune system. Therapies such as immune checkpoint inhibitors depend not only on tumour genetics, but also on the presence, location and functional state of immune cells within the tumour microenvironment. Spatial biology has enabled us to identify regions of immune suppression, uncover molecular pathways driving tumour progression and discover new therapeutic targets.
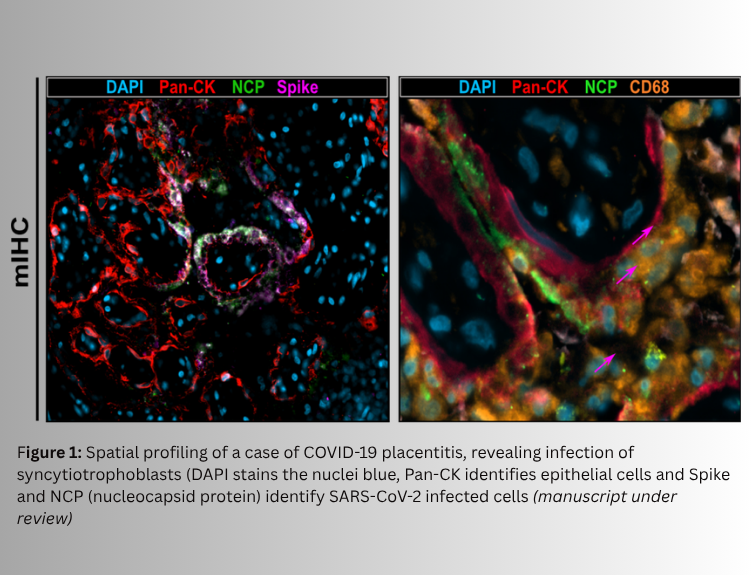
Recent research by our group has also demonstrated how powerful these technologies can be in understanding disease mechanisms. For example, we combined spatial transcriptomics (a powerful molecular profiling technique that maps gene expression within intact tissue sections) and proteomics to study the lung tissues of patients who died from severe COVID-19, enabling us to produce maps detailing the complexity of COVID-19 infections [1]. This work demonstrated that virus-infected lung epithelial cells were surrounded by immunosuppressive macrophages expressing immune checkpoint proteins, while antiviral T-cells were relatively depleted in the immediate microenvironment of infected cells. Moreover, in rare cases in which SARS-CoV-2 infects the placenta we have been able to define that this occurs in specific cells that line the placental tissues (syncytiotrophoblasts; Figure 1) Thus, these spatial insights are helping us explain why anti-SARS-CoV-2 viral immune responses fail in people who succumb to COVID-19.
Importantly, the same study also revealed that collagen deposition in lung tissue was linked to disease severity and could be detected in the blood as a biomarker, serving as a predictor of patient outcomes. Such discoveries highlight the translational potential of spatial biology: interrogating complex tissue architectures can lead directly to clinically useful biomarkers that help stratify patients and guide treatment decisions. For pathology as a discipline, these developments represent an important evolution rather than a replacement of traditional methods. The microscope remains central to diagnosis, but it is now complemented by powerful molecular technologies that provide deeper insights into disease biology. The pathologist of the future will increasingly work at the intersection of histology, molecular biology and computational analysis, interpreting tissue morphology as well as the complex spatial networks that define disease.
As spatial technologies continue to advance, they promise to reshape both research and clinical practice. By mapping the cellular ecosystems of cancer and other diseases, spatial biology is helping move pathology from descriptive observation toward mechanistic understanding and precision medicine, ultimately improving how we diagnose, stratify and treat patients. To discuss potential research collaborations with Professor Paul Murray, please get in touch via email at pamurray@rcsi-mub.com.
Reference



